Full article · 7 min read
Tardigrade Genomes and Shield Proteins: How Tiny Water Bears Protect Their Cells
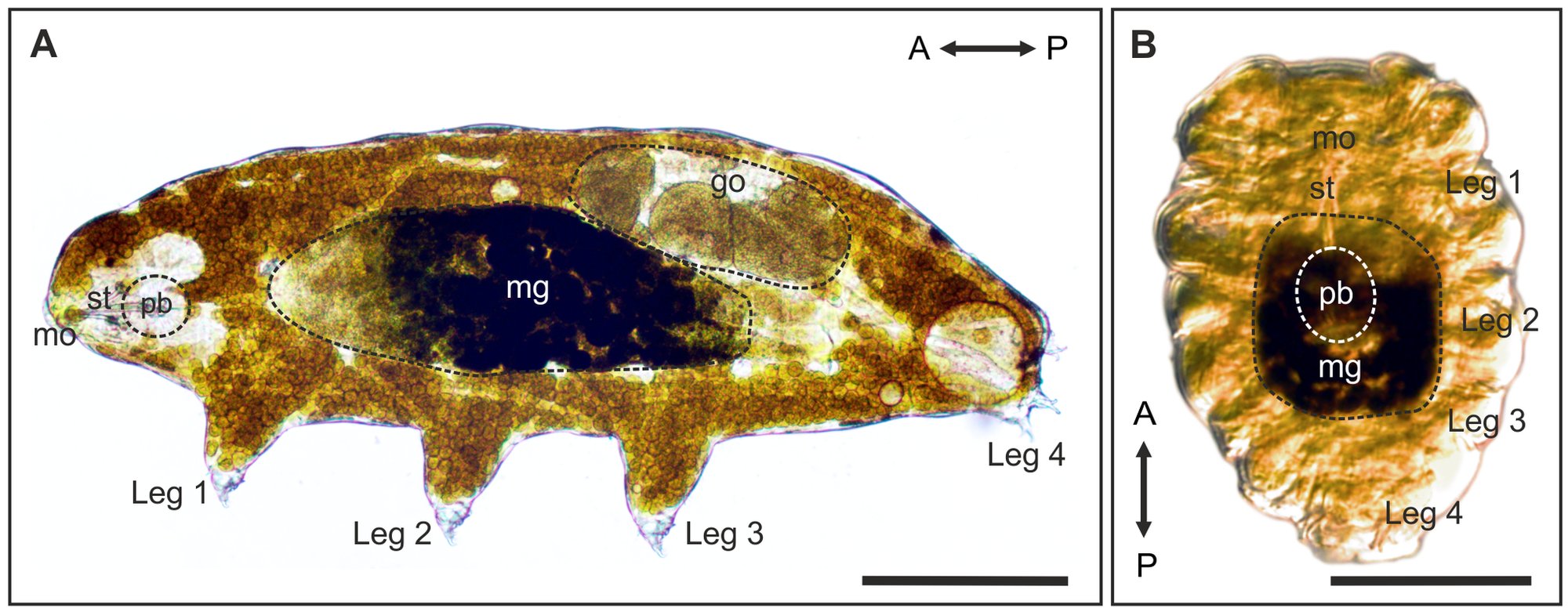
Tardigrades are famous for surviving conditions that would quickly kill most animals, but some of the most intriguing clues to that toughness are hidden in their genomes and protective proteins. These microscopic animals, also called water bears, are usually only about 0.5 mm long when fully grown, yet they have become important model organisms for studying how living cells can endure drying, radiation, freezing, and other stresses.
Two tardigrade species stand out in this story: Hypsibius exemplaris and Ramazzottius varieornatus. They are both useful for genomic research, but they highlight slightly different sides of tardigrade biology. One has a compact, practical genome for laboratory work, while the other is known as one of the most stress-tolerant tardigrades studied so far.
Two small genomes with big scientific value
Tardigrade genomes vary widely in size, but the two species featured here show just how compact these genomes can be.
Hypsibius exemplaris, part of the Hypsibius dujardini group, has a genome of about 100 megabase pairs. A megabase is one million DNA letter-pairs, so that means its genome contains about 100 million of those paired units. It also has a generation time of about two weeks, can be cultured indefinitely, and can be cryopreserved. That makes it especially useful in research, because scientists can maintain it in the lab over long periods.
Ramazzottius varieornatus has an even smaller genome, about 55 megabase pairs, roughly half the size of the genome of H. exemplaris. It is also described as one of the most stress-tolerant tardigrade species. This combination of a small genome and extraordinary tolerance makes it especially attractive for scientists trying to connect genes with survival under severe conditions.
Genomic research on tardigrades is not just about counting DNA. It also helps scientists reconstruct the evolution of the tardigrade body plan and understand how these animals became miniaturised from larger ancestors. Tardigrades lack several of the Hox genes found in arthropods. Hox genes are developmental genes that help organize the body layout of an animal. In tardigrades, a large intermediate region of the body axis is missing compared with arthropods. In insects, that missing region would correspond to the entire thorax and abdomen. As a result, nearly the whole tardigrade body, except the last pair of legs, corresponds to segments homologous to the head region in arthropods.
The remarkable Dsup protein
Among tardigrade protective molecules, one of the most talked about is Dsup, short for damage suppressor. This protein helps protect tardigrade DNA from radiation damage.
DNA in cells is packaged around protein structures called nucleosomes. You can think of nucleosomes as spools around which DNA is wound. The Dsup proteins of Ramazzottius varieornatus and Hypsibius exemplaris promote survival by binding to these nucleosomes and protecting chromosomal DNA from hydroxyl radicals.
Hydroxyl radicals are highly reactive molecules that can damage DNA. Because they react so readily, they can break or alter genetic material, which can be fatal or harmful to cells. Dsup acts like a molecular shield, helping reduce that damage.
The Dsup protein of R. varieornatus has another notable feature: it confers resistance to ultraviolet-C by upregulating DNA repair genes. Ultraviolet-C, or UV-C, is the most energetic and DNA-damaging part of ultraviolet radiation mentioned here. Upregulating DNA repair genes means the protein helps increase the activity of genes involved in repairing damaged DNA.
This matters because tardigrades are known to withstand ionising radiation and even survive exposure to outer space under some conditions. In a 2007 experiment, dehydrated tardigrades were exposed for 10 days to vacuum or to both vacuum and solar ultraviolet during the FOTON-M3 mission. More than 68% of the individuals protected from ultraviolet were reanimated by rehydration once back on Earth, and many produced viable embryos. Later, tardigrades also flew on the International Space Station on STS-134, where they showed they could survive microgravity and cosmic radiation. Protective mechanisms such as Dsup help explain why tardigrades are so interesting to biologists.
Tardigrades are not “gene hoarders”
One idea that attracted attention was whether tardigrades might owe their unusual toughness to having picked up huge numbers of genes from other organisms. The data from Ramazzottius varieornatus do not support that dramatic picture.
About 1.6% of the genes in the genome of R. varieornatus are the result of horizontal gene transfer from other species. Horizontal gene transfer means acquiring genes from non-parent organisms rather than inheriting them directly through normal reproduction. In simple terms, it is like borrowing bits of DNA from other species.
But that 1.6% does not imply any dramatic effect. In other words, the genome of this tardigrade does not look like a bizarre collection of foreign genes stitched together to create its resilience. Its survival abilities are better understood as arising from its own biology, including its specialized protective proteins and its responses to stress.
Surviving drying out: CAHS, LEA, and other protective proteins
One of the most important challenges for many tardigrades is desiccation, meaning extreme drying out. Terrestrial and freshwater tardigrades can tolerate long periods without water by entering cryptobiosis, a suspended state in which metabolic activity stops. In this state, they pull in their legs and form a desiccated cyst called a tun.
In the tun state, tardigrades can go without food or water for several years. They also become highly resistant to stresses including lack of oxygen, vacuum, ionising radiation, high pressure, and temperatures from as low as −272 °C to as much as +149 °C, at least for short periods.
For a long time, scientists thought this ability depended on high levels of trehalose, a sugar often associated with surviving desiccation in other organisms. But tardigrades do not synthesize enough trehalose for that function. Instead, they produce intrinsically disordered proteins in response to desiccation.
Intrinsically disordered proteins do not keep one rigid, stable shape like many other proteins do. That flexibility appears to be important for protection during drying. Three of these proteins are specific to tardigrades and are called tardigrade specific proteins. These proteins may protect membranes from damage by associating with the polar heads of lipid molecules. Lipids are fat-like molecules that make up cell membranes, and their polar heads are the water-attracting parts facing outward.
These proteins may also form a glass-like matrix that protects the cytoplasm during desiccation. Cytoplasm is the material inside the cell that surrounds its internal structures. A glass-like matrix suggests a stabilizing network that helps hold cellular components in place while water is absent.
The molecular response is extensive. In Hypsibius exemplaris, anhydrobiosis in response to desiccation involves 1,422 genes being upregulated during the process. Of those, 406 are specific to tardigrades, 55 are intrinsically disordered proteins, and the others are globular proteins with unknown functions. Anhydrobiosis is the form of life suspension associated with surviving almost complete drying.
Researchers see particular promise in proteins such as CAHS and LEA. CAHS stands for Cytoplasmic Abundant Heat Soluble proteins, while LEA stands for Late Embryogenesis Abundant proteins. These proteins are being studied for their ability to protect against desiccation, and some CAHS proteins could help prevent programmed cell death, also called apoptosis. Apoptosis is the controlled self-destruction process cells use when they are damaged or no longer needed.
Why this matters for biomedicine
The protective systems of tardigrades are of interest to biomedical research because they suggest new ways to defend cells from injury.
Dsup has potential because of its ability to protect against damage, especially damage involving DNA. CAHS and LEA proteins are promising because they may help protect cells and cellular components from desiccation. If scientists can understand exactly how these proteins work, they may eventually develop new tools for preserving biological materials or reducing stress-related cell damage.
That interest comes directly from what tardigrades already do naturally. These animals are not considered true extremophiles in the strict sense, because they are not adapted to thrive in many extreme environments. Instead, they are exceptional at enduring them. Their survival still drops the longer exposure lasts, but their capacity to withstand severe stress remains extraordinary.
Tiny animals, outsized impact
Tardigrades are made of only about 1000 cells, yet they continue to reshape how scientists think about stress tolerance, DNA protection, and the limits of animal survival. Their compact genomes, unusual body plan, and distinctive proteins make them more than just a curiosity under the microscope.
Hypsibius exemplaris offers a practical model with a compact 100-megabase genome and laboratory-friendly traits. Ramazzottius varieornatus adds an even smaller genome, exceptional stress tolerance, and the famous Dsup protein that helps guard DNA. Together, they show that biological resilience does not require a giant genome or huge numbers of borrowed genes. Sometimes, a tiny animal with the right molecular toolkit is enough to challenge assumptions about what life can endure.
Sources
Based on information from Tardigrade.
More like this
Give your brain a damage-suppressor boost — download DeepSwipe and armor up with tiny facts that pack a huge punch.