Full article · 9 min read
Biology: How Life Uses Energy
Life is powered by a constant flow of energy. From a muscle cell using fuel during exercise to a plant leaf turning sunlight into sugar, living things survive because their cells can capture, convert, store, and spend energy in controlled ways.
At the center of this story are a few core processes: cellular respiration, ATP production, fermentation, and photosynthesis. Together, they explain how organisms keep themselves alive, grow, respond to their environments, and help shape the planet itself.
Why cells need energy at all
All cells require energy to sustain cellular processes. The full set of chemical reactions in an organism is called metabolism. Metabolism has three main purposes: converting food into energy to run cellular processes, converting food or fuel into building blocks such as monomers, and eliminating metabolic wastes.
These reactions are guided by enzymes, which are molecules that act as catalysts. In simple terms, a catalyst helps a chemical reaction happen faster without being used up. Enzymes also lower the activation energy needed for reactions, allowing cells to carry out processes that would otherwise happen too slowly or not at all under normal conditions.
Metabolism includes two broad kinds of reactions. Catabolic reactions break compounds down and usually release energy. Anabolic reactions build compounds up and usually consume energy. Cellular respiration is one of the most important catabolic processes because it allows cells to extract usable energy from nutrients.
Cellular respiration: controlled fire inside the cell
Cellular respiration is the set of reactions and processes that convert chemical energy from nutrients into adenosine triphosphate, or ATP. ATP is the molecule cells use to power many of their activities, which is why it is often described as the cell’s energy currency.
Respiration also releases waste products. The reactions involved are catabolic, meaning they break larger molecules into smaller ones while releasing energy. Although cellular respiration is technically a combustion reaction, it does not look like a flame. In a cell, the energy is released gradually through a series of biochemical steps rather than all at once.
That slow, controlled release is what makes respiration so powerful for life. Instead of wasting energy as a sudden burst, cells capture it bit by bit and use it to make ATP.
Sugar in the form of glucose is the main nutrient used by animal and plant cells in respiration. When oxygen is involved, the process is called aerobic respiration.
ATP: the usable energy packet of the cell
ATP stands for adenosine triphosphate. Its importance comes from its role as an immediate source of usable energy for cellular processes. Cells do not simply burn food directly every time they need to do work. Instead, they convert energy from nutrients into ATP, which can then power activities throughout the cell.
Mitochondria are key players in this process in eukaryotic cells. These organelles generate ATP to power cellular processes. In aerobic respiration, some major stages occur in the cytoplasm, but crucial later stages take place in the mitochondria, including steps in the mitochondrial matrix and on the mitochondrial cristae, the folded inner membrane structures that support ATP-producing reactions.
The four stages of aerobic respiration
Aerobic respiration has four stages: glycolysis, the citric acid cycle, the electron transport chain, and oxidative phosphorylation.
Glycolysis
Glycolysis takes place in the cytoplasm. In this process, glucose is converted into two pyruvates, and two net ATP molecules are produced.
Pyruvate is a smaller molecule produced by breaking down glucose. It serves as a key intermediate, meaning it is an important stop along a larger metabolic pathway.
Formation of acetyl-CoA and the citric acid cycle
Each pyruvate is then oxidized into acetyl-CoA by the pyruvate dehydrogenase complex. This step also generates NADH and carbon dioxide.
Acetyl-CoA enters the citric acid cycle, which takes place inside the mitochondrial matrix. By the end of the cycle, the total yield from one glucose molecule, or two pyruvates, is 6 NADH, 2 FADH2, and 2 ATP molecules.
NADH and FADH2 are molecules that carry high-energy electrons. Those electrons become crucial in the next phase of respiration.
Electron transport chain and oxidative phosphorylation
The final major ATP-producing stage is oxidative phosphorylation. In eukaryotes, this occurs in the mitochondrial cristae. It includes the electron transport chain, a series of four protein complexes that pass electrons from one complex to another.
As electrons move through this chain, energy is released. That energy is used to pump protons, which are hydrogen ions, across the inner mitochondrial membrane. This builds a proton motive force, a stored energy gradient across the membrane.
That gradient then powers the enzyme ATP synthase, which synthesizes more ATP by phosphorylating ADP. In plain language, ATP synthase is a molecular machine that uses the proton gradient to help produce ATP.
At the end of the chain, molecular oxygen serves as the final electron acceptor. This role is essential in aerobic respiration. Without oxygen to accept those electrons, the chain cannot continue in the same way.
Why oxygen matters so much
Oxygen’s job as the final electron acceptor is one of the most important details in energy biology. During aerobic respiration, electrons are passed along the electron transport chain until they are finally accepted by oxygen.
That final handoff keeps the system running. Because of it, cells can continue releasing energy from NADH and FADH2, maintain the proton motive force, and make large amounts of ATP through oxidative phosphorylation.
This is why oxygen is so tightly connected with high-efficiency energy production in many forms of life.
When oxygen disappears: fermentation steps in
If oxygen is not present, pyruvate is not metabolized by cellular respiration. Instead, it undergoes fermentation. In this situation, pyruvate remains in the cytoplasm and is converted into waste products that can be removed from the cell.
Fermentation serves a crucial purpose: it oxidizes NADH to NAD+, allowing glycolysis to continue. NAD+ is a helper molecule the cell needs in order to keep breaking down glucose through glycolysis.
Without oxygen, fermentation prevents NADH from building up in the cytoplasm and restores NAD+ so the cell can keep producing at least some ATP through glycolysis.
This is a much more limited energy strategy than aerobic respiration, but it can keep cells going when oxygen is unavailable.
Lactic acid fermentation
In skeletal muscles, the waste product of fermentation is lactic acid. This process is called lactic acid fermentation. During strenuous exercise, when energy demands exceed energy supply, the respiratory chain cannot process all the hydrogen atoms joined by NADH. During anaerobic glycolysis, NAD+ is regenerated when pairs of hydrogen combine with pyruvate to form lactate.
This reaction is catalyzed by lactate dehydrogenase and is reversible. Lactate can also be used as an indirect precursor for liver glycogen. During recovery, when oxygen becomes available, NAD+ attaches to hydrogen from lactate to form ATP.
Alcoholic fermentation
In yeast, fermentation produces ethanol and carbon dioxide. This is known as alcoholic or ethanol fermentation. The ATP generated in fermentation is made by substrate-level phosphorylation, which does not require oxygen.
Photosynthesis: storing sunlight as chemical energy
If respiration explains how cells release energy from nutrients, photosynthesis explains how much of that energy enters the living world in the first place.
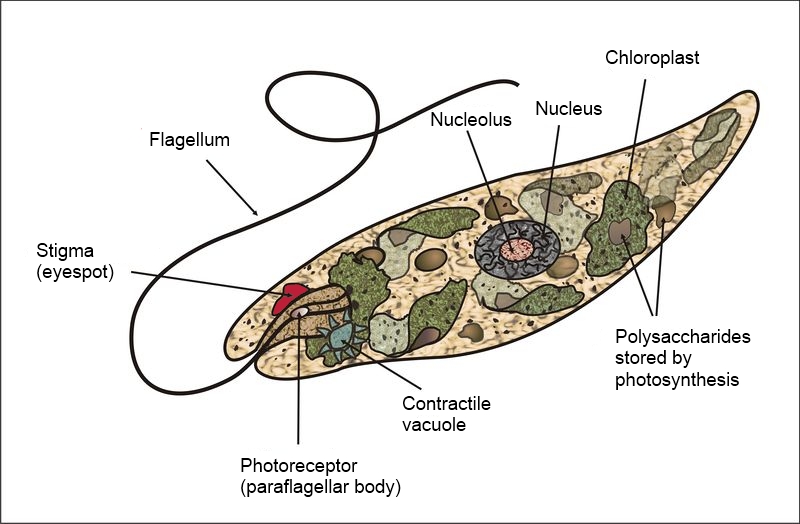
Photosynthesis is the process used by plants and other organisms to convert light energy into chemical energy. That chemical energy is stored in carbohydrate molecules such as sugars, which are synthesized from carbon dioxide and water. In most cases, oxygen is released as a waste product.
Most plants, algae, and cyanobacteria perform photosynthesis. This process is largely responsible for producing and maintaining the oxygen content of Earth’s atmosphere, and it supplies most of the energy necessary for life on Earth.
That means photosynthesis does two extraordinary things at once: it stores energy in sugars and helps make aerobic respiration possible by releasing oxygen.
The stages of photosynthesis
Photosynthesis has four stages: light absorption, electron transport, ATP synthesis, and carbon fixation.
Light absorption
The first step is light absorption. Light energy is absorbed by chlorophyll pigments attached to proteins in the thylakoid membranes.
Electron transport
The absorbed light energy removes electrons from water and transfers them to a primary electron acceptor, a quinone designated as Q. The electrons then move through a series of electron carriers until they reach a final electron acceptor, usually the oxidized form of NADP+, which is reduced to NADPH in photosystem I.
At the same time, electron transport is coupled to the movement of protons from the stroma to the thylakoid membrane space, creating a pH gradient.
ATP synthesis
During the third stage, protons move down their concentration gradient through ATP synthase. That movement is coupled to ATP synthesis.
Carbon fixation
The NADPH and ATP produced in the earlier stages then help drive the synthesis of glucose by fixing atmospheric carbon dioxide into existing organic carbon compounds such as ribulose bisphosphate in the Calvin cycle.
The result is stored chemical energy in sugar molecules, which can later be used in metabolism and cellular respiration.
How photosynthesis and respiration connect
These two processes are deeply linked. Photosynthesis captures light energy and stores it in sugars. Cellular respiration breaks down those sugars to release energy in a usable form, mainly ATP.
Photosynthesis also releases oxygen, and aerobic respiration depends on oxygen as the final electron acceptor. Together, these processes help explain the energy flow of life on Earth.
Energy from the sun enters ecosystems through photosynthesis and is incorporated into plant tissue. By feeding on plants and on one another, animals move matter and energy through ecosystems. Decomposers then break down dead organic matter, releasing carbon back to the atmosphere and helping cycle nutrients.
The energy foundation of life on Earth
From mitochondria producing ATP to plants releasing oxygen, the biology of energy ties together cells, organisms, and entire ecosystems. It explains how life sustains itself moment by moment and how the atmosphere and food webs of Earth are maintained.
Cellular respiration shows how cells extract energy in a controlled way. Fermentation reveals how life can adapt when oxygen is absent. Photosynthesis shows how light becomes food and how oxygen enters the atmosphere.
In short, life runs on energy transformations. And every breath, bite, and leaf is part of that ongoing biological exchange.
Sources
Based on information from Biology.
More like this
Charge up your curiosity like a mitochondrion — download DeepSwipe and turn spare moments into ATP for your brain.